Volunteer Management
Our volunteer management infrastructure is built around three principles: safety, dignity, and transparency. Every volunteer who participates in a Lazent Clinical study is supported by trained clinical staff from first screening through study completion and follow-up.


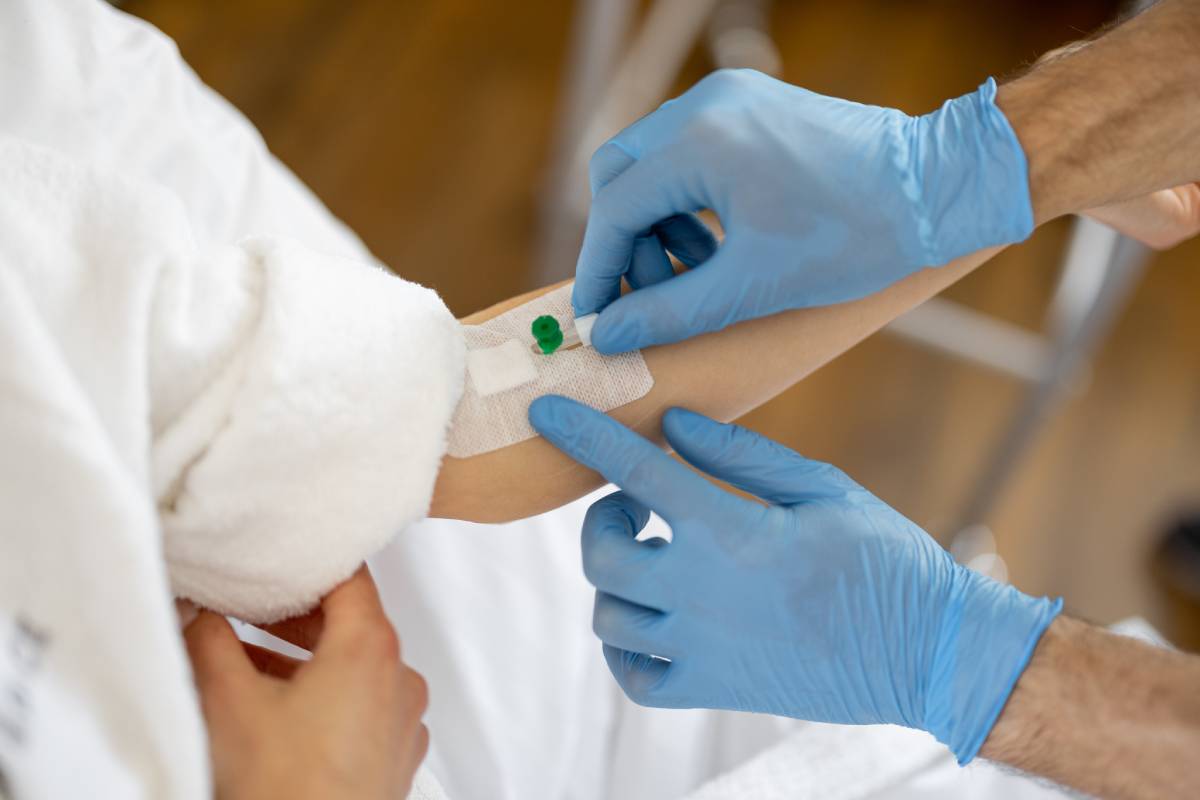
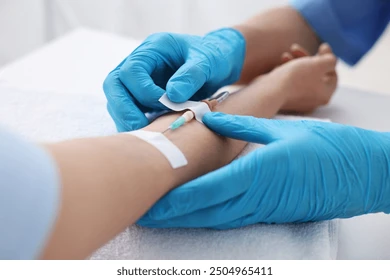
Clinical Facility
The Lazent Clinical facility spans a fully equipped clinical pharmacology unit with dedicated zones for ICU monitoring, bed-based confinement, dosing, sample handling, and volunteer welfare. Each area is designed to GCP standards.

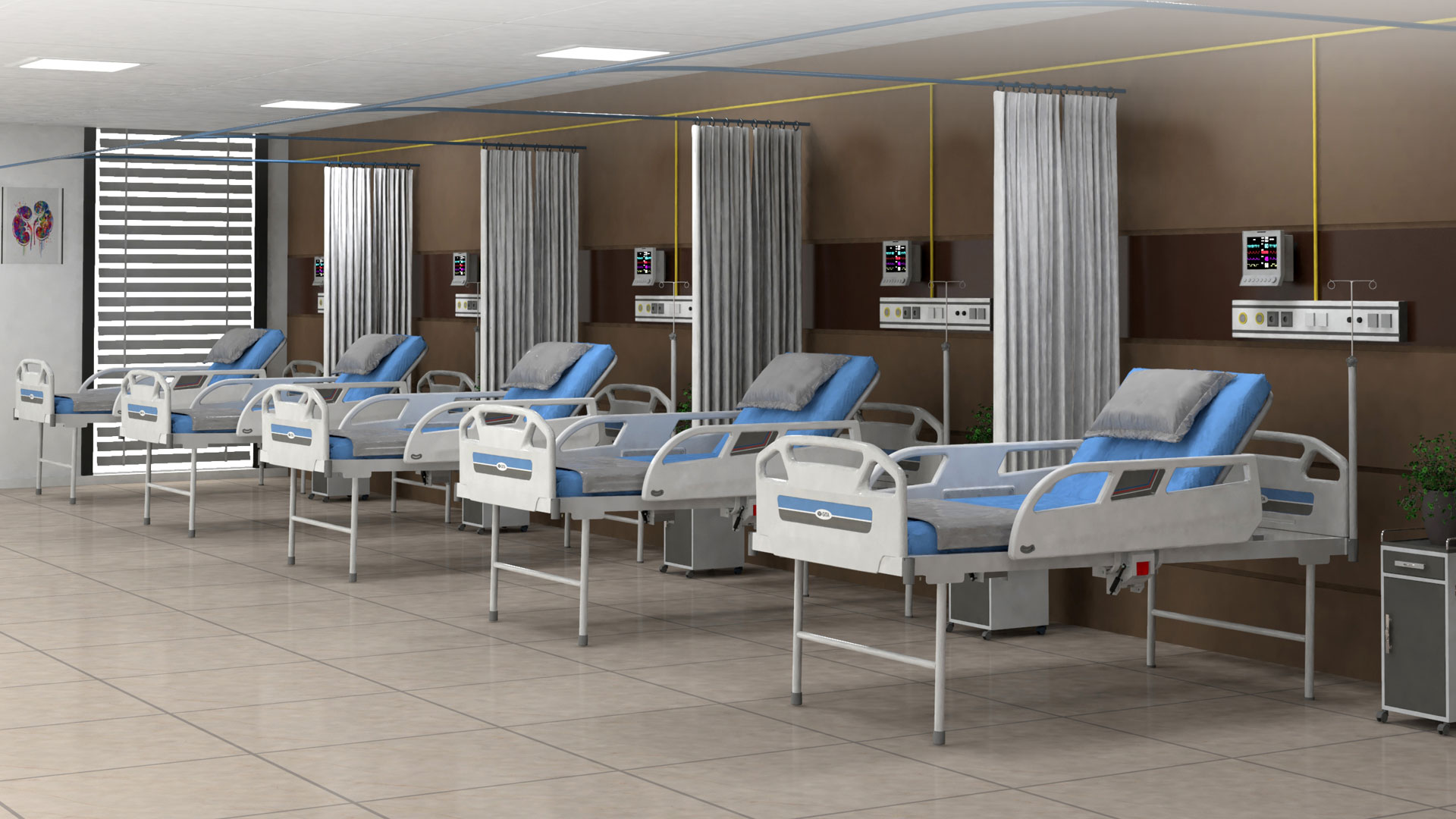


Bioanalytical Laboratory
State-of-the-art LC-MS/MS instruments operated under GCLP compliance for validated regulatory-grade bioanalytical data.




Sample Storage & Cold Chain
Temperature-controlled storage systems ensuring sample integrity from collection through analysis.



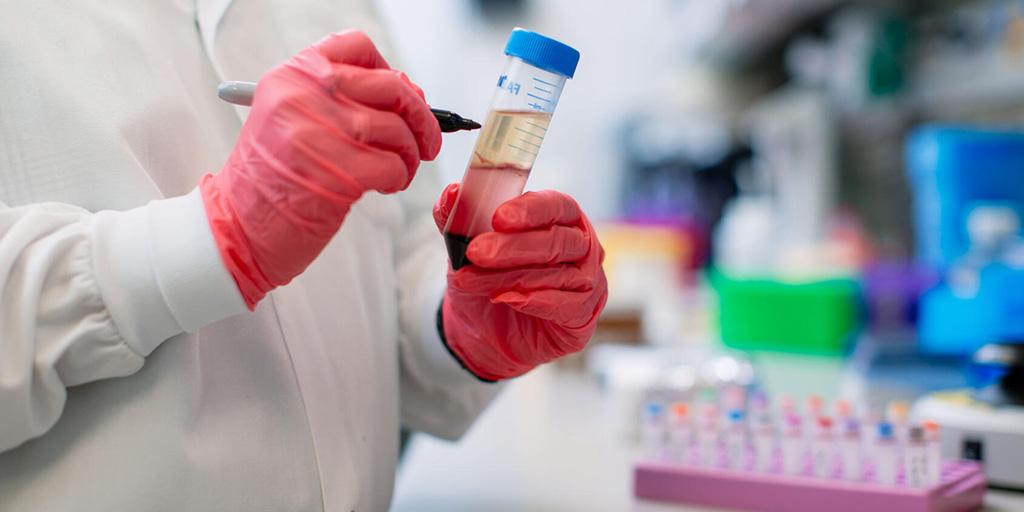
Medical Consultation & Research

